季碳手性中心广泛存在于天然产物、药物和农用化学品中,是增强分子三维复杂性的关键结构单元。然而,受限于严重的空间位阻与有限的构象灵活性,这类手性中心的构建难度极大,在脂肪族体系中的合成尤为困难。这限制了药物发现及对新化学空间的探索。因此,开发高效、通用的对映选择性构建季碳手性中心的方法,已成为现代合成化学领域的热点。
近日,四川大学华西药学院陈芬儿院士/王慧婧副教授团队通过基因挖掘,成功鉴定出一种来源于Ruegeria sp. R14的新型环己酮单加氧酶RuCHMO。该酶对4,4-二取代环己酮底物表现出良好的催化活性和>99%的对映选择性。在此基础上,团队采用FRISM策略对酶进行半理性设计,获得了双突变体Mu2(F490M/L201I)。该突变体对多种功能化底物的催化效率显著提升,分离产率最高可达97%,对映选择性普遍超过99%。基于此,提出了“酶促不对称Baeyer-Villiger反应构筑手性中心 + 化学转化延伸骨架”的组合策略,实现了天然产物(+)-Kopsiyunnanine K的化学酶法全合成。
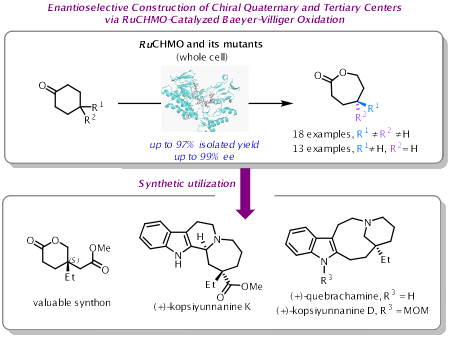
该研究不仅丰富了酶促不对称Baeyer-Villiger反应的催化工具箱,也为复杂手性季碳中心的构建提供了绿色、高效的酶催化剂。研究成果体现了酶工程与化学合成的深度融合,展示了交叉学科策略在解决合成难题中的潜力。未来,该策略有望拓展至更多复杂天然产物及药物分子的合成中。相关成果发表于ACS Catalysis(https://doi.org/10.1021/acscatal.5c08950)。该研究得到了国家自然科学基金的资助。我院2025级博士生张敏为论文第一作者,王慧婧副教授与陈芬儿院士为共同通讯作者。
Chen and Wang’s Group Achieves Breakthrough in Asymmetric Baeyer–Villiger Oxidation
Quaternary stereocenters are key structural motifs in natural products, pharmaceuticals, and agrochemicals, adding three-dimensional complexity to molecular structures. However, constructing these centers,especially in aliphatic systems,remains challenging due to severe steric hindrance and limited conformational flexibility. This bottleneck has hindered drug discovery and the exploration of new chemical space, making the development of efficient, enantioselective methods a high-priority goal in modern synthesis.
Recently, Fen-Er Chen and Huijing Wang’s groupidentified a novel cyclohexanone monooxygenase, RuCHMO, fromRuegeriasp. R14 through genome mining. This enzyme showed promising catalytic activity and >99% enantioselectivity toward 4,4-disubstituted cyclohexanones. Using a FRISM-based semi-rational engineering approach, the team developed a double mutant, Mu2 (F490M/L201I), which exhibited significantly improved catalytic efficiency across a range of functionalized substrates, achieving isolated yields up to 97% and consistently >99%ee.Leveraging this enzyme, the team developed a combinatorial strategy,“enzymatic asymmetric Baeyer–Villiger oxidation to establish stereocenters followed by chemical transformations to extend the carbon skeleton”,and completed the chemoenzymatic total synthesis of the natural product (+)-kopsiyunnanine K.
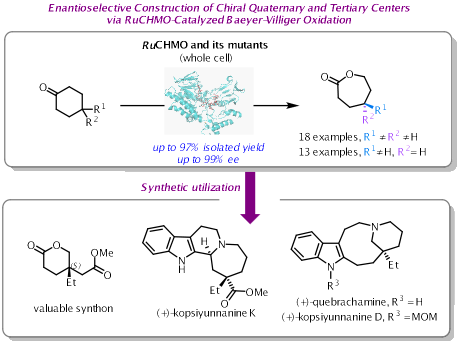
This work expands the catalytic toolbox for enantioselective Baeyer–Villiger oxidations and provides a green, efficient enzymatic platform for constructing complex quaternary stereocenters. It exemplifies the power of integrating enzyme engineering with chemical synthesis to tackle long-standing challenges in asymmetric synthesis. The strategy holds promise for application to a wide range of complex natural products and pharmaceuticals.
The findings are published inACS Catalysis(https://doi.org/10.1021/acscatal.5c08950). The research was supported by the National Natural Science Foundation of China. Min Zhang, a 2025 Ph.D. candidate at the college, is the first author, with Associate Professor Huijing Wang and Academician Fen-Er Chen serving as co-corresponding authors.
